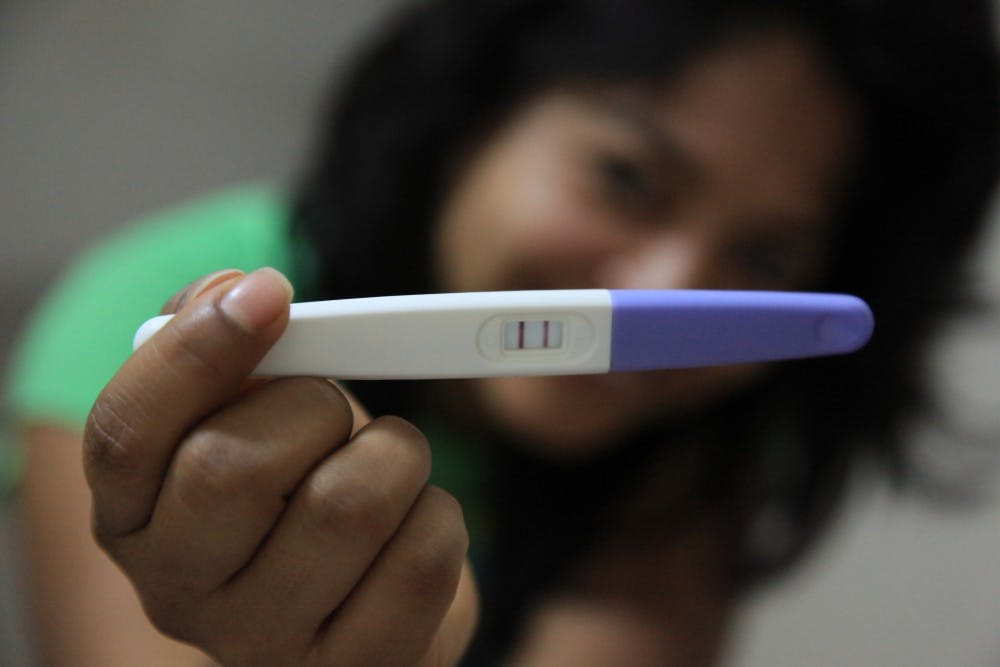
Two Penn graduates have just created the first FDA-approved flushable pregnancy test — with a 99 percent accuracy rate, TechCrunch reported.
This is the latest product initiative from Lia Diagnostics, a company co-founded by 2014 Engineering Ph.D. graduates Bethany Edwards and Anna Couturier Simpson.
Edwards is a 2006 graduate from Temple University’s Fox School of Business and Simpson is a 2011 graduate of Tyler School of Art. They met while completing graduate studies at Penn through the Integrated Product Design program, which is a joint degree through The School of Engineering and Applied Science, The Wharton School, and The School of Design.
Edwards conceived the idea for Lia Diagnostics after graduation, and, along with Simpson, officially founded Lia Diagnostics in Dec. 2014.
“What we’ve done here is essentially creating a new category of water-dispersable, biodegradable diagnostics,” Edwards said of the paper compostable test in the Lia Diagnostics pitch to venture capitalists and judges at Disrupt Berlin. "This is just the start for us."
The U.S. Drug and Food Administration granted pre-market approval for the pregnancy test on Nov. 20, and Lia Diagnostics will begin selling its product on its website and Amazon starting mid-2018. The test will cost between $9 and $22, a similar price to conventional pregnancy tests.
In 2015, Lia Diagnostics won Wharton’s Social Innovator Start-Up competition. The company was featured on Philly Magazine’s 2017 Startups To Watch list, as well as on Philadelphia Business Journal’s “15 women who are changing the face of tech in the region” list in 2016.
Most recently, Lia Diagnostics traveled to TechCrunch’s Disrupt Berlin Startup Battlefield and beat out more than 14 other companies to win the grand prize of $50,000 on Dec. 5, Philadelphia Magazine reported.
RELATED:
Penn named one of the best schools in the country for producing entrepreneurs
At PennApps XVI, students made inter-dimensional robots and hung out with the founder of Quora
“Single-use diagnostics are only used for a couple of minutes, and they’re all made out of plastic and non-sustainable materials,” Edwards told TechCrunch, adding that although Lia offers a non-biodegradable test alternative, it still works the same way.
The company sees itself as “changing the face of reproductive healthcare at the pharmacy and beyond,” according to the website.
The diagnostics company is backed by investments by Ben Franklin Technology Partners and Dreamit Health, and is part of the Digital Health Accelerator at the University City Science Center in Philadelphia, according to MedCity News.